Legal & Regulatory
FDA approvals, reimbursement, litigation, and policy
158 articles

Legal & Regulatory
SpinePoint Receives U.S. FDA 510(k) Clearance for Flex-Z™ Cervical Cage / First Product in the Ultra-Low Stiffness Cushioning Interbody Platform
halvorsonMar 28, 2026
1 views

Legal & Regulatory
Spinal Elements Expands Ventana® Platform with FDA 510(k) Clearance and First Cases of Ventana® A ALIF System
halvorsonMar 19, 2026
1 views

Legal & Regulatory
2nd FDA Clearance for Novel Instrument Sterilization System
elizabethhofheinzmedmphMar 2, 2026
1 views
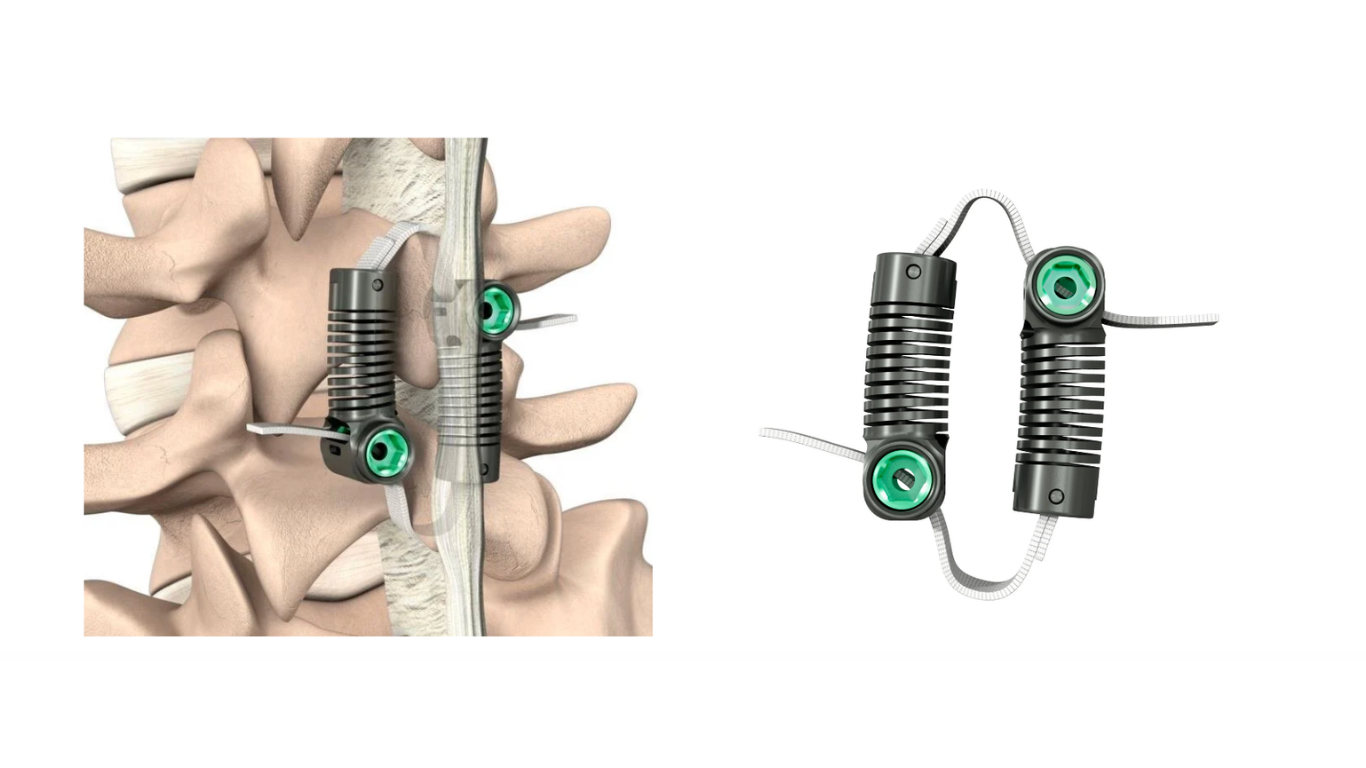
Legal & Regulatory
FDA Approves (PMA) Dynamic Sagittal Tethering for Spondy
elizabethhofheinzmedmphMar 2, 2026
1 views

Legal & Regulatory
Synergy Spine Solutions® Receives FDA Approval for its Synergy Disc®, Expanding Cervical Disc Replacement Options for U.S. Patients
halvorsonFeb 27, 2026

Legal & Regulatory
4WEB Medical Receives 510(k) Clearance to Market its New SI Joint Truss System™
halvorsonFeb 25, 2026

Legal & Regulatory
MiRus® Receives FDA 510(k) Clearance for IO™ Expandable Wedge Osteotomy System
halvorsonFeb 24, 2026

Legal & Regulatory
FDA Clears IST’s ONE TRAY® for 365 Day Storage, Expanding Sterilization Flexibility for Surgical Facilities
halvorsonFeb 20, 2026
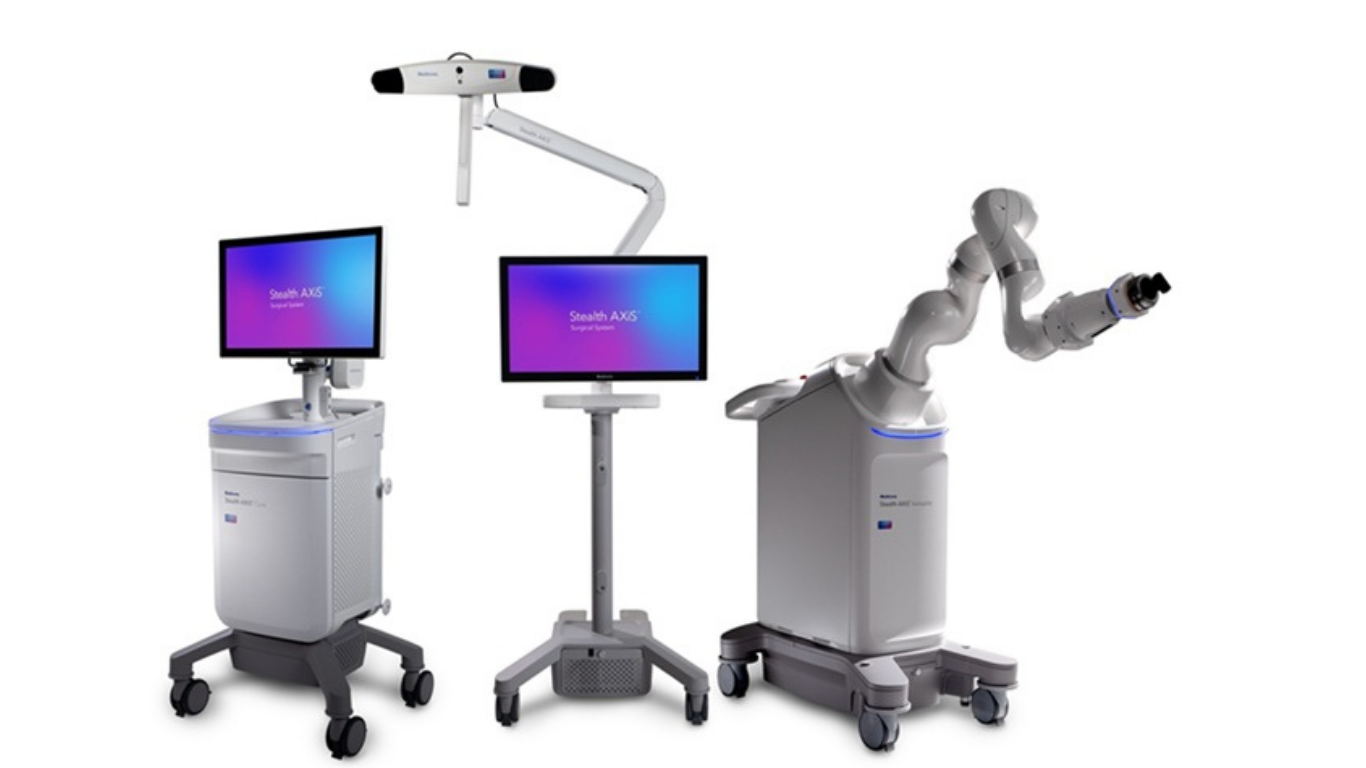
Legal & Regulatory
Medtronic’s Next Gen Intelligent Spine Platform Unveiled
elizabethhofheinzmedmphFeb 19, 2026

Legal & Regulatory
Medtronic Announces FDA approval of Infuse™ bone graft for one-and-two level Transforaminal Lumbar Interbody Fusion (TLIF) spine procedures
halvorsonFeb 17, 2026

Legal & Regulatory
Medtronic receives FDA clearance for Stealth AXiS™ surgical system, first integrated planning, navigation and robotics platform for spine surgery
halvorsonFeb 13, 2026

Legal & Regulatory
Patent Granted for Artificially Intelligent Surgical Planning
elizabethhofheinzmedmphFeb 13, 2026
Page 1 of 14