Premarket approvals are rare and come with substantially more clinical support than 510(k) clearances, so when the rare PMA is granted, it worth noting and in this case, celebrating.
The FDA has granted Premarket Approval to the LimiFlex™ Dynamic Sagittal Tether — a motion-preserving stabilization system designed for lumbar spinal stenosis associated with Grade I degenerative spondylolisthesis following decompression.
This is the long-awaited middle ground solution that lives between decompression alone and instrumented fusion.
Decompression Versus Instability
Decompression relieves the stenosis…but what about the instability? Fusion solves the instability…but at the cost of motion — and potentially adjacent segment stress.
Degenerative spondylolisthesis accounts for more than 250,000 procedures annually in the U.S. and roughly half of lumbar fusions. For decades, fusion has been the default “insurance policy” against postoperative instability and recurrence.
But what if stabilization didn’t require rigid fixation?
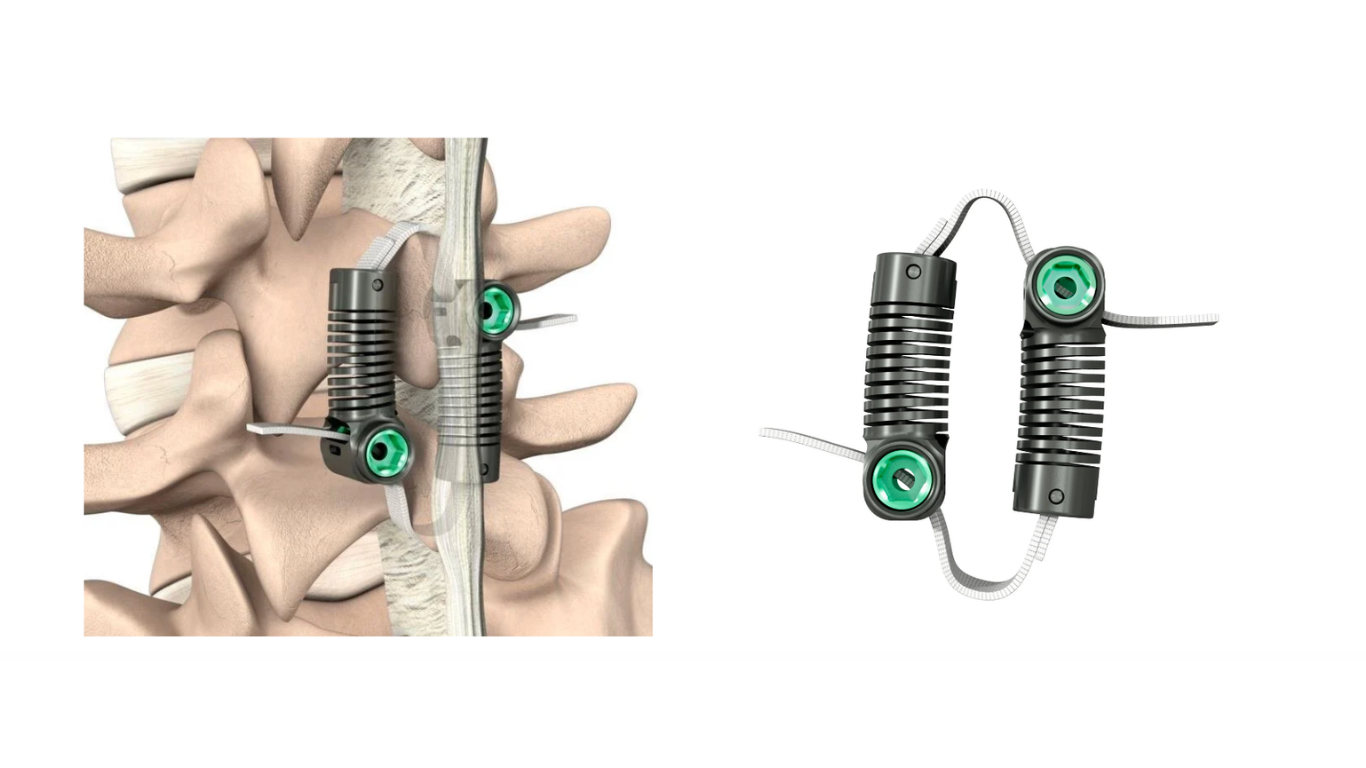
Enter dynamic saggital tethering, brand named LimiFlex.
What is it, exactly?
Developed by Silicon Valley-based Empirical Spine and rooted in research from Stanford University School of Medicine, LimiFlex provides dynamic restraint of pathologic motion after decompression — without eliminating segmental movement.
Think of it as controlled stability rather than rigid immobilization.
Instead of reconstructing the segment, the system stabilizes while preserving native anatomy and physiologic motion. Surgically, it aligns more closely with decompression in magnitude than with full reconstruction.
Shorter cases. Potential outpatient pathway. Motion preserved.
That’s a combination that tends to get surgeons’ attention.
Equivalent Outcomes to Fusion, Way Shorter Procedure Times
In the FDA pivotal investigational device exemption trial (IDE), LimiFlex demonstrated:
- Non-inferior 2-year outcomes compared with instrumented fusion
- Substantially shorter procedures
- Support for outpatient treatment in appropriate patients
That’s not a small statement. Non-inferior to fusion — without fusing — represents a meaningful shift in the treatment landscape.
Surgeons Weigh In
Todd Alamin, M.D., co-founder and Professor of Orthopaedic Spine Surgery at Stanford, summed it up succinctly: patients can now experience symptom relief “without the added physiologic and mechanical burden of fusion.”
Hyun Bae, M.D. of Cedars-Sinai Medical Center called LimiFlex “one of the most thoughtful advances in spine care,” noting what we all know: patients strongly prefer motion-preserving and outpatient options.
Rick Sasso, M.D. of Indiana Spine Group reported that nearly all investigational device exemption patients at his center were treated in an ambulatory surgery setting — and would otherwise have undergone fusion.
That’s a real-world shift in surgical magnitude.
Why This Is Interesting for Orthopedic Surgeons
Whether you’re a fellowship-trained spine surgeon or an orthopedic surgeon who keeps an eye on motion-preserving innovation, this approval represents something bigger.
We are officially entering a new stabilization category: Decompression → Dynamic Stabilization → Fusion
For years, that middle lane has been more theoretical than mainstream. Premarket approval changes that conversation.
What happens next?
According to Empirical Spine leadership, an initial limited U.S. launch will begin at select investigational device exemption centers, followed by broader expansion later this year to establish additional centers of excellence.
As with any new technology, long-term durability, adoption curves, and real-world outcomes will ultimately define its role.
But for now?
Spine surgeons have a new option — one that challenges the long-standing assumption that instability automatically equals fusion.
And that makes this approval more than just another device clearance. It’s a meaningful step in the ongoing evolution of motion-preserving spine care.