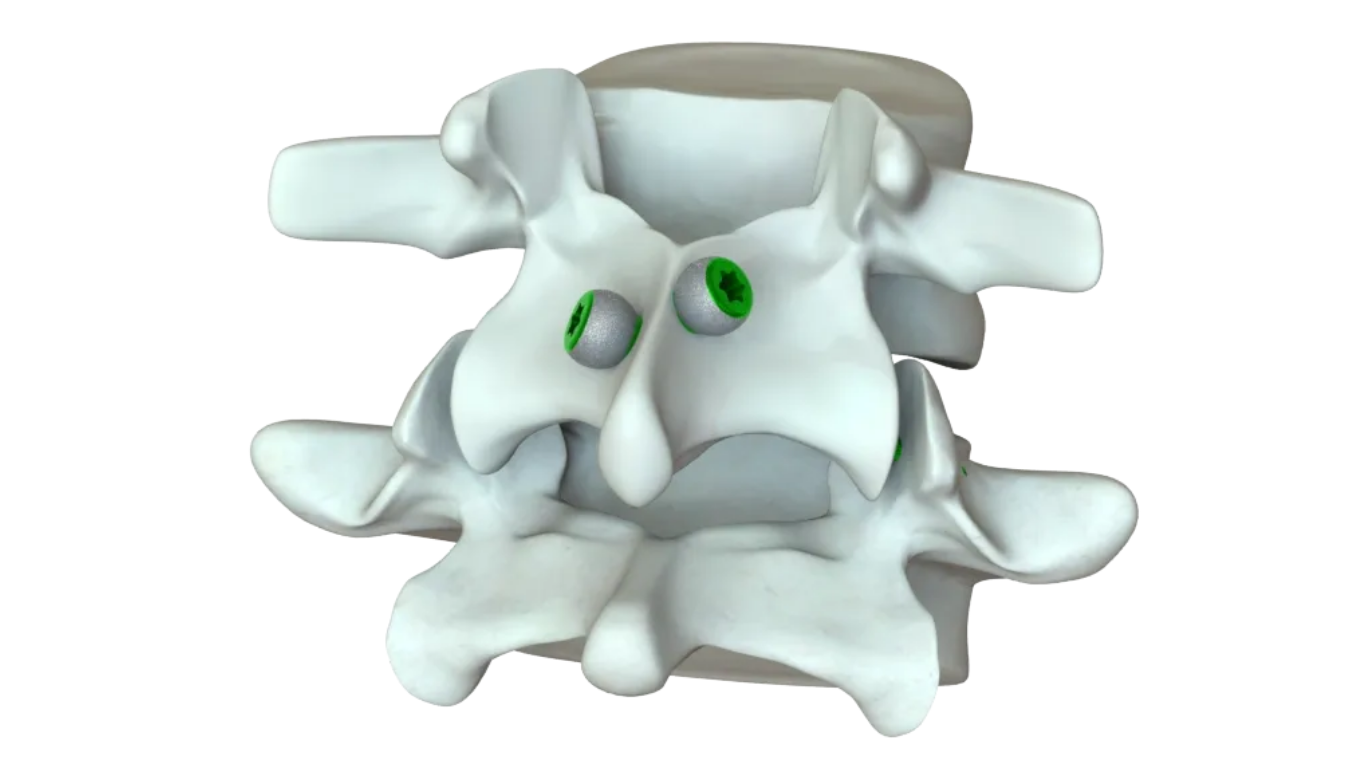
Bountiful, UT – March 23, 2026—VySpine, a spine innovation leader using differentiated materials and designs, announced today that it has received 510(k) clearance from the FDA for the its DiversiVy™ Facet Screw System which is designed for use in the lumbosacral spine to stabilize the spine and aid in fusion.
The DiversiVy™ Facet Screw System is intended to stabilize the spine as an aid to fusion by transfacet fixation. The device is indicated for posterior surgical treatment with or without bone graft, at single or multiple levels, of any or all of the following spinal levels L1 to S1 (inclusive): Spondylolisthesis, Spondylolysis, Pseudoarthrosis or failed previous fusions which are symptomatic: Degenerative Disc Disease (DDD) as defined by back pain of discogenic origin with degeneration of disc confirmed by history and radiographic studies and/or degenerative disease of the facets with instability.
“The DiversiVy™ clearance is the latest in a long line of Vy Spine implants to provide physicians with optimal options for their patients.” Bret Berry, President of Product Development for Vy Spine notes. “Vy Spine is focused on expanding our portfolio of products with an emphasis on simple and robust fixation, at a price conscious value.”